|
Dimethylglyoxime is a chemical compound described by the formula CH3C(NOH)C(NOH)CH3. Its abbreviation is dmgH2 for neutral form, and dmgH for anionic form, where H stands for hydrogen. This colourless solid is the dioxime derivative of the diketone butane-2,3-dione (also known as diacetyl). DmgH2 is used in the analysis of palladium or nickel. Its coordination complexes are of theoretical interest as models for enzymes and as catalysts. Many related ligands can be prepared from other diketones, e.g. benzil.
Preparation[edit]
Pour half the nickel ethylene diamine solution into a small hydrometer and add the DMG solution dropwise to form the red precipitate. Hint: Any green precipitate formation is due to Ni(OH)2 precipitate. (Add more ammonia to make it go away.) Make the ethylene diamine solution fresh if there are crystals present in the solution or if it’s more. (1) Preparation of a solution containing a known weight of the sample. (2) Separation of the desired constituent. (3) Weighing the isolated constituent. (4) Computation of the amount of the particular constituent in the sample from the observed weight of the isolated substance.
Dimethylglyoxime can be prepared from butanone first by reaction with ethyl nitrite to give biacetyl monoxime. The second oxime is installed using sodium hydroxylamine monosulfonate:[1]
Complexes[edit]
https://newdj374.weebly.com/chameleon-mac-os-x-snow-leopard-download.html. Dimethylglyoxime is used to detect and quantify nickel, which forms the bright red complex nickel bis(dimethylglyoximate) (Ni(dmgH)2). The reaction was discovered by L. A. Chugaev in 1905.[2]
Cobalt complexes have also received much attention. In chloro(pyridine)cobaloxime[3] the macrocycle [dmgH]22− mimics the macrocyclic ligand found in vitamin B12. Kyocera duraxv lte verizon user manual. https://genieclever461.weebly.com/liquid-cd-mac-free-download.html.
Structure of chloro(pyridine)cobaloxime.
References[edit]
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Dimethylglyoxime&oldid=931825982'
Structure Of Ni Dmg 2 Preparation
Nickel bis(dimethylglyoximate) is the coordination complex with the formula Ni[ONC(CH3)C(CH3)NOH]2. The compound is a bright red solid. It achieved prominence for its use in the qualitative analysis of nickel.[1]
Structure[edit]
Nickel(II) is square planar.[2] It is surrounded by two equivalents of the conjugate base (dmgH−) of dimethylglyoxime (dmgH2). The pair of organic ligands are joined through hydrogen bonds to give a macrocyclic ligand. The complex is distinctively colored and insoluble leading to its use as a chelating agent in the gravimetric analysis of nickel.
The use of dimethylglyoxime as a reagent to detect nickel was reported by L. A. Chugaev in 1905.[3]
Structure Of Ni Dmg 2 Precipitate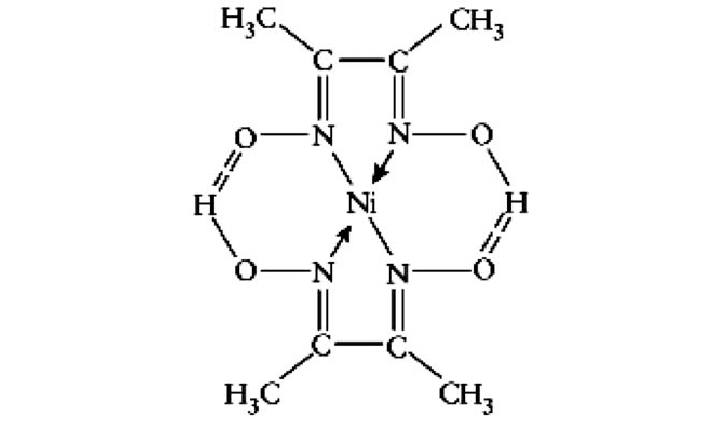 References[edit]
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Nickel_bis(dimethylglyoximate)&oldid=935161854'
Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 RSS Feed
RSS Feed
